


Delta Research Partners was founded in 2003. Now with 2 locations in Bastrop, La and Monroe, La. Delta is an independent research center dedicated to conducting high-quality clinical research trials in multiple therapeutic areas. Delta has a strong network represented by our Principal Investigator and several Sub-Investigators, all board-certified in Internal Medicine and Gastroenterology. Our team is lead by Principal Investigator, Dr. Raj Bhandari, who is triple-board certified in Internal Medicine, Gastroenterology, and Nutrition.
With a combination of over 30 years of experience in gastroenterology and liver disease research, our providers at DRP are focused on the treatment and early detection of GI and liver disease. We strive to educate the community and provide resources to our fellow practioners to better detect and manage GI & liver disease while it is still in its preventative stages. When choosing DRP, you are choosing to make a difference in the future of GI & liver related medicine. Study participants can expect:
Delta is located in Northeast Louisiana with an estimated 350,000+ population in our community and surrounding areas. Our own database consists of over 100,000 subjects. We also have an extensive referring physician database with over 350 physicians that are a great source to our subject recruitment. The physicians are from various specialties including:
Delta is committed to assisting physicians and their subjects to having access to new and promising treatments that can improve quality of life. Our community has a positive attitude toward research and we are able to generate a tremendous response with the use of IRB approved ads and flyers.
Delta is affiliated with two independent Gastroenterology Practices in Northeast Louisiana and an Ambulatory Surgery Center (ASC). Since we have two (2) separate study sites (Monroe & Bastrop), monitors can monitor two different sites with one visit.

A FibroScan is an exam of your liver that is similar to an ultrasound. It uses vibration controlled, transient elastography technology to determine your liver health non-invasively. The test takes approximately 10 minutes and provides immediate results.
Any patient with one or more of the following risk factors is at a greater risk for developing fatty liver disease. Fatty liver disease does not have symptoms, making risk factors even more helpful in determining who should get screened.
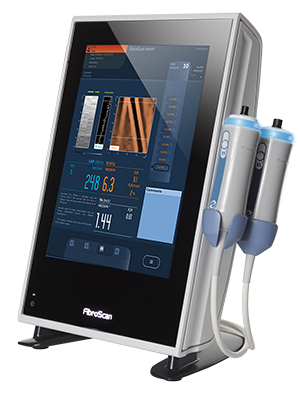
When other tests indicate that your liver may not be working properly, your doctor may want to do a fibroscan to more accurately learn the condition of your liver. Unlike a liver biopsy, a fibroscan is quick, painless and performed in a clinic setting.
A Fibroscan helps:
To have a Fibroscan you will not be able to eat or drink anything for three hours before the test. You may not have a Fibroscan exam if you have any or all of the following:
Fibroscan is a simple exam that involves you lying on your back with your right arm raised behind your head. The technician will apply a water-based gel to your skin on the ride side of your body at the rib cage and then apply a probe with slight pressure. This is not painful. The exam includes ten consecutive measurements taken at the same location.
After the exam you may eat and drink as usual and continue your daily activities. There is no down time or recovery period needed after a fibroscan. A study investigator or physician will interpret the fibroscan report and review the results with you, as well as provide you personalized lifestyle modifications and recommendations that you can take with you to follow up with your PCP.
